In present study β-Cyclodextrine and Hydroxy Propyl- β-Cyclodextrine were used as the water soluble carriers for increasing the solubility of Ziprasidone using lyophilisation technique. The complexes were prepared using various techniques physical mixing, kneading, co-precipitation, solvent evaporation, spray drying and freeze drying. They form a water soluble complex with the drug having very low solubility. Complexing with cyclodextrin is one of the most promising techniques to enhance the solubility of poor water soluble moiety. The oral bioavailability is only 39-60% with a t 1/2 of about 6 hours (Approximately). Therefore it shows poor oral bioavailability. The oral absorption of Ziprasidone is limited and it reaches maximum concentration within 6-7 hours. It is a pinkish brown coloured powder having very low solubility in water (21.12 mg/L). It mainly acts as antagonist of D2 dopamine and 5HT 2A (serotonin, 5HT, 5-hydroxytryptamine) receptors. This makes it a unique special choice of antipsychotic agent. It has its unique multi receptor binding affinity.
Ziprasidone Hydrochloride (HCl) is one of the newly introduced atypical antipsychotic. Due to the economic and therapeutic efficacy of second generation antipsychotics these are preferred mostly for the treatment than first generation antipsychotics. INTRODUCTION: 1-7 The way of treating psychosis was totally changed when atypical antipsychotics were introduced in the therapy of psychosis. Keywords:īioavailability, Inclusion complex, Lyophilisation, Ziprasidone HCl In the catalepsy study the selected inclusion complex showed increase in bioavailability compared to the drug and almost all the data obtained from study was found to be 99.99% significant with the control. Therefore the freeze dried complex with HP-β-CD was selected for Catalepsy study on Wistar rats. During in vitro study result obtained that the lyophilized complexes with HPβ-CD showed 100% drug release within 10 min were as the lyophilized complexes with β– CD showed 100% drug release in 25 min. The inclusion complexes were characterized using DSC and XRD technique. The saturation solubility analysis demonstrated highest increase in the solubility of drug after complexation with HP-β-CD by lyophilisation technique. All the inclusion complexes prepared by lyophilisation technique showed remarkable increase in the solubility compared to the pure Ziprasidone HCl. The β-Cyclodextrine and Hydroxy Propyl- β-Cyclodextrine were used as the water soluble carriers for increasing the solubility of Ziprasidone. The main purpose of this study is to enhance the solubility of Ziprasidone HCl using lyophilisation technique. It is pinkish brown colored powder having very low solubility in water (21.12 mg/L).

It mainly acts as antagonist of D 2 dopamine receptors as and 5HT 2A (serotonin, 5HT, 5-hydroxytryptamine) receptors. It has its own unique multi receptor binding affinity.

PES Modern College of Pharmacy 2, Nigdi, Pune - 411044, Maharashtra, India.ĪBSTRACT: Ziprasidone HCl is a newly introduced atypical antipsychotic drug. Marathwada Mitra Mandal’s College of Pharmacy 1, Thergaon (Kalewadi), Pune - 411033, Maharashtra, India. SOLUBILITY ENHANCEMENT OF BCS CLASS II DRUG USING LYOPHILISATION TECHNIQUE AND DETERMINATION OF BIOAVAILABILITY IN ANIMALS USING CATALEPSY MODEL
#Bcs class 4 drugs list full
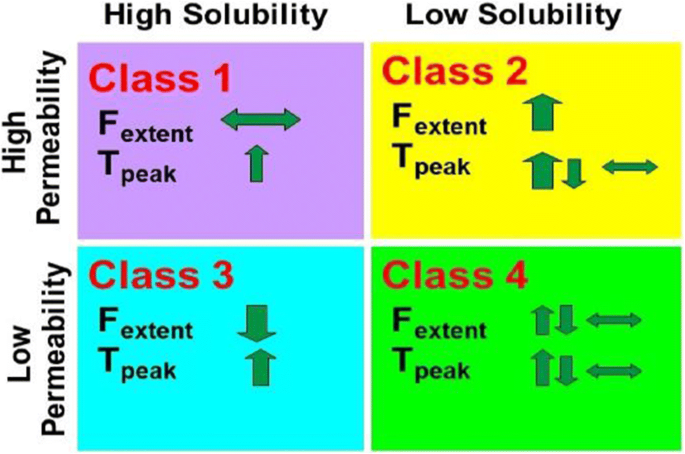
SOLUBILITY ENHANCEMENT OF BCS CLASS II DRUG USING LYOPHILISATION TECHNIQUE AND DETERMINATION OF BIOAVAILABILITY IN ANIMALS USING CATALEPSY MODEL HTML Full Text The WHO EML was used as it is a publicly available list of Class 1, 2, 3, and 4 drugs. We evaluated 263 approved generic drugs of IR products listed on the WHO EML to find out the distribution of BCS Class 1, 2, 3, and 4 drugs in approved ANDA applications during the 2000 to 2011 period. Biopharmaceutics Classification System (BCS) has provided a mechanistic framework for understanding the concept of drug absorption in terms of permeability and solubility. Twenty-one (84%) of these belong to class I (highly soluble, highly permeable), 10 (17%) to class II (poorly soluble, highly permeable), 24 (39%) to class III (highly soluble, poorly permeable) and 6 (10%) to class IV (poorly soluble, poorly permeable). Of the 130 orally administered drugs on the WHO list, 61 could be classified with certainty.
